 For example, the line spectra shown below for the elements helium and carbon are clearly quite different (colors can be seen in the PDF document on-line). The result is called a line emission spectrum, and can serve as a ‘fingerprint’ of the element to which the atoms belong. If emitted photons are in the visible region of the spectrum, they may be perceived as lines of different colors (note that photons outside the visible spectrum may also be emitted, but cannot be seen). The spacing between energy levels in an atom determines the sizes of the transitions that occur, and thus the energy and wavelengths of the collection of photons emitted. 
However, when electrons subsequently return from higher energy levels to lower energy levels, energy is released predominantly in the form of electromagnetic radiation. The energy absorbed could be in the form of heat (as in flame tests), or electrical energy, or electromagnetic radiation. 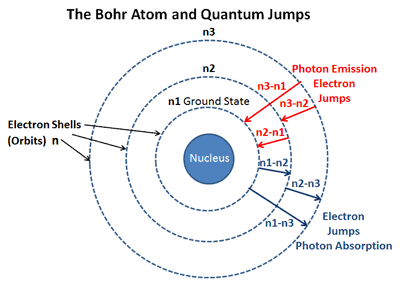
So, how does electromagnetic radiation relate to flame tests? Well, when an atom or ion absorbs energy, its electrons can make transitions from lower energy levels to higher energy levels. Other examples of electromagnetic radiation include X-rays, ultraviolet light, infrared light, microwaves and radio waves. Differences in the wavelengths of visible light are manifested as different colors, shown in the color spectrum below (colors can be seen in the PDF document on-line). Visible light is the most familiar example of electromagnetic radiation. For instance, hydrogen is the simplest atom and thus, it has the simplest spectrum.J \cdot s\). The frequencies of the light emitted by a particular element exhibit a regular pattern.Emission Line spectrum is when an atomic gas or vapor is excited at low pressure by passing an electric current through it, the emitted radiation has a spectrum that contains specific wavelengths only.The atomic spectra can be defined as the spectrum of the electromagnetic radiation that is emitted or absorbed by electrons during transitions between different energy levels within an atom.The first Atomic structure model was proposed by J.Important Notes On Atoms Important Questions The empirical formulas are given for the observed wavelengths, series such as Lyman, Paschen, Brackett and Pfund and Balmer formula in terms of frequency of light are important as the wavelengths give that hydrogen atom radiate or absorb but these results are empirical and don’t give any reasoning as to why only certain frequencies are observed in the hydrogen spectrum.The Balmer formula when written in terms of frequency of light is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
